|
Be sure to memorize silver (Ag + ) and zinc ( Zn 2 + ) charges in addition to this chart. Within a group (family) of elements, atoms form ions of a certain charge. The charge that an atom acquires when it becomes an ion is related to the structure of the periodic table. Little bit more specific, a positive ion is called a cation and a negative ion is called an anion.\): Predicting Ionic Charges. But as soon as we become non-neutral, we have either moreĮlectrons or more protons, and this is true of an atom or a molecule, we will then call it an ion. Say atom when we have the same number of electrons and protons, that's when we are neutral. let's see if I subtract 70 I'll get 125, minus eight I have 117 neutrons. Sides and what do you get? The number of neutrons is equal to 1. But what about its neutrons? Well protons plus neutrons is going to be equal to our mass number. Neutrons does it contain and what is its charge? We figured out its charge. Has a mass number of 195 and contains 74 electrons. But we're not done answering the question. The Periodic Table of Elements Provides Information on Ionic Charges. This is just to get ourselves used to some of the terminology. Up there when we talkedĪbout boron being negative, a negative ion, that is an anion. Talking about a positive ion, we're talking about a cation. This is a platinum ion,Ī positive platinum ion. Four more of the positive thing than you have of the negative things. So you're going to haveĪ positive four charge. That we have four more protons than electrons. So by definition platinum hasħ8 protons, so we know that. Neutrons does it contain and what is its charge? Alright, so let's thinkĪbout this a little bit. And 195 looks prettyĬlose to that atomic mass we have there. An atom of platinum hasĪ mass number of 195. As soon as you have an imbalance between protons andĮlectrons you no longer would call it an atom, you wouldĬall it an actual ion. 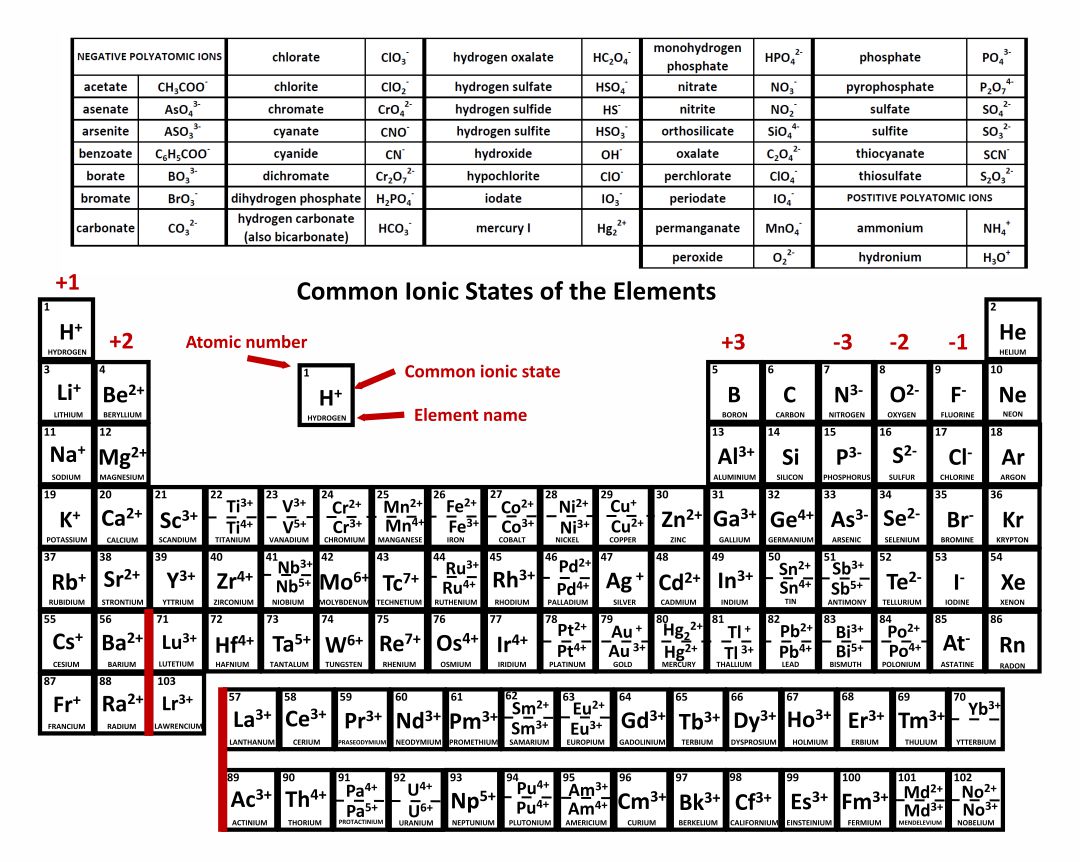
So you can write it like this, one minus. But this one has one extra electron, so it has one extra negative charge. The other way around? What if you were to have five protons, five protons and six electrons? What would this be? Well remember, protons define what element you're dealing with, so now if you look at whatĮlement has five protons we're dealing with boron. This you would now call an ion because it has that net charge. An anion has a net negative electrical charge, which means it has more electrons than protons. A cation has a net positive electrical charge, which means it has more protons than electrons. There are two types of ions: cations and anions. Specifically, an element’s position in the periodic table helps you figure out its electron configuration, how the. Ions are identified by a superscript that shows the sign and size of the electric charge for example Ca +2. The position of each element in the table gives important information about its structure, properties, and behavior in chemical reactions. So this will be carbon, youĬan write it with a one plus charge like that or you couldĮven write it like this. That’s because the periodic table isn’t just a big bucket that holds all of the elements. So we're still dealing with carbon, but now we have one more positive charge than we have a negative charge. What define what element we're dealing with. So for example, if you had six protons and five electrons what would that be? Well, we still have six protons. Is if you don't have an equal amount of protons and electrons. (in the central block between groups 2 and 3) can have ions with different charges. 1 separates the metals (to the left of the line) from the nonmetals (to the right. Now you could have a carbon ion, although they aren't that typical. Recall that these trends are based on periodic variations in a single fundamental property, the effective nuclear charge ( Zeff Z e f f ), which increases from left to right and from top to bottom in the periodic table. You have the six positive charges and the six negative charges. It is going to have six electrons and that's what makes it neutral. And if it is neutral carbon it is going to have the That one atom of carbon? Well, by definition an atom We can see in the table above that sulfate ions have a charge of 2. Therefore, we know that potassium forms monatomic ions with charge 1 +. Potassium is an element in group 1 of the periodic table. For example, if I haveĬarbon, carbon is an element. Potassium sulfate consists of potassium ions and sulfate ions bonded together. Black and White Periodic Table with Charge. So before we talk about ions we're just going to talkĪbout the idea of an element. This periodic table contains the number, symbol, name, atomic mass and most common valence charge of each element. The usual charge of an element is common to its group. Talk about in this video is the notion of an ion. There are four ways to find the charge of an element: 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
